Enhance your pre-clinical data package with our transformational IndEx-2 system
Our rapidly customisable in vitro cell line platform can be used to determine antigen density activation thresholds of targeted candidate immunotherapies in a cost-effective manner.
In addition, IndEx-2 can be easily integrated with immune-oncology primary cell assays, so it can provide a complete picture of a targeted drug candidate’s mechanism of action and safety profile.
Get in touch with our team to learn how you could apply this platform to the pre-clinical discovery of any antibody-targeted therapy; CAR-engineered cell therapies, bispecific antibodies, multispecifics, immune cell engagers, monoclonal antibodies, ADCs and more.
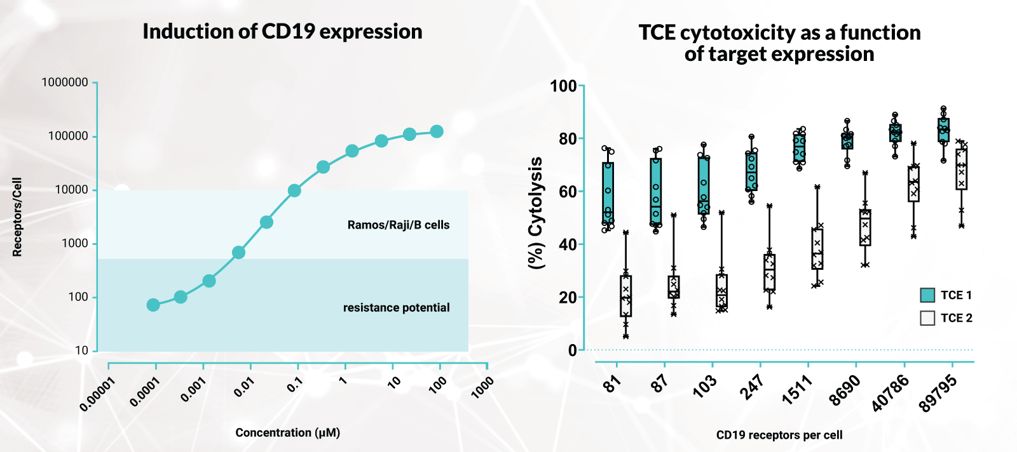
Left hand figure: Dose dependent induction of CD19 expression. CHO-K1 cells containing an inducible CD19 construct. Inducer was added at the indicated concentrations and the cells were incubated for 48 hours. CD19 expression was detected by flow cytometry analysis and the number of CD19 receptors per cell was quantified using a Quantibrite PE bead fluorescence quantification kit (BD Biosciences). The CD19 receptors per cell for Ramos, Raji (diseased) and B cells isolated from PBMCs (healthy) are shown by way of comparison.
Right hand figure: Use of CHO-K1 inducible CD19 cells for the comparison of the cytotoxicity of two CD19xCD3 bispecific T cell engagers (TCEs) across a range of CD19 receptor expression levels. CHO-K1 inducible CD19 cells were seeded in xCELLigence RTCA plates and treated with a concentration series of inducer for 24 hours to induce CD19 expression. CD8+ T cells were isolated from donor PBMCs (n=10) and incubated with target cells at a 3:1 ratio in the presence of 225pM CD19xCD3 TCE 1 or TCE 2. Target cell death, as determined by impedance loss due to cell detachment, was determined after a further 72 hours. Data show mean of donors (n=10) +/- SD.
How to apply
IndEx-2
This powerful cell-based system can be used;
To titrate the expression of one, or two, TAOIs over a range of biologically relevant expression levels. Model pathological and physiological expression levels.
To determine the therapeutic window of targeted therapies in the same cell line background. No confounding genetic background differences.
Determine the activation threshold or minimum number of receptors for therapeutic efficacy. Select candidates with lower chance of antigen escape.
For the pre-clinical evaluation of risks associated with ‘on-target/off-tumour’ toxicities.
Currently available inducible cell lines
Our pre-developed single-inducible (CHO-K1) IndEx-2 cell lines are ready to use in assays now:
CD19
CD22
Her2
BCMA
PD-L1
FOLR1
EpCAM
CEACAM5
B7H3
PSMA
We now have two additional cell lines in different cancer cell backgrounds to use in assays:
A549 - FOLR1
HCT116 - B7H4
Why choose Antibody Analytics' in vitro IndEx-2 system?
One of the key features of IndEx-2 is that it's titratable, so it is capable of a finely controlled induction of a nominated target antigen of interest (TAOI). Additionally, it has a large dynamic range of TAOI expression in any genetic background which makes it the perfect tool to test a variety of antibody-targeted therapies. And above all, independently controlled expression of one, or two TAOIs makes IndEx-2 an exceptional model.
Collaborative approach
Our in-house teams of molecular biologists and immunologists have the expertise to create custom cell lines and assays with your challenge in mind. We design in consultation with you.
Tuned expression levels
Pick a specific level of receptor expression to model disease states more representatively or help determine drug activation thresholds.
Integrated expertise
The cell line can be used immediately in our range of bioassays and translational immunology assays. This provides you with a more comprehensive understanding of mechanism of action and functionality.
Watch our CSO's presentation on IndEx-2 associated assays
The number of antibody-targeted I-O therapeutics in development is growing year after year. However, there persists a lack of in vitro systems to interrogate the efficacy and safety of these modalities.
In response to this, we have developed IndEx-2, an advanced in vitro cell-based platform which allows the expression of one, or two, target antigens over a range of biologically relevant levels, facilitating the determination of the precise antigen density activation thresholds of targeted candidate immunotherapies and antibody-based therapeutics.